東京理科大学 工学部工業化学科HiMat-Chemical Engineering


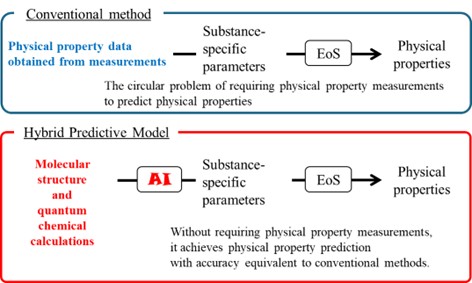
Hybrid Physical Property Prediction Model Combining “Equation of State” and “AI”
An equation of state (EoS) is used to correlate and predict various physical properties across a wide range of temperature and pressure conditions, from pure substances to mixtures. This is because it can determine not only the pressure-volume-temperature relationship but also various thermodynamic state variables. The equation of state typically taught in high school chemistry is PV=nRT. However, this is the ideal gas equation of state and cannot be applied to real substances. Therefore, numerous types of equations have been proposed to accommodate real substances. These equations of state are incorporated and used within process simulators, such as Aspen+ and PROII, which are essential tools for process design.
Calculations using a equation of state applicable to actual substances involve obtaining substance-specific parameters from measured physical properties, applying them to the equation of state, and predicting physical properties. This creates a circular problem where predicting physical properties requires known physical properties. To address this issue, we are developing a novel model that combines AI with an integrated “equation of state and AI” model to predict physical properties without requiring prior knowledge of those properties.
Peng-Robinson equation of state-van der Waals one fluid mixing rule, interaction parameters kij, lij between CO2 and organic solvents
Peng-Robinson equation of state
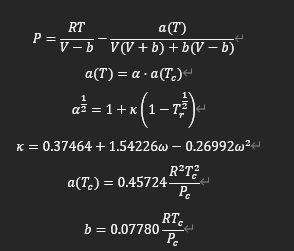
van der Waals one fluid mixing rule
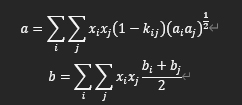
- Prediction model based on critical points and eccentricity factors of organic solvents
- Prediction model based on molecular information obtained from quantum science computing (future update)
- Prediction model based on molecular information of organic solvents in carbon dioxide using PCM theory (future update)
